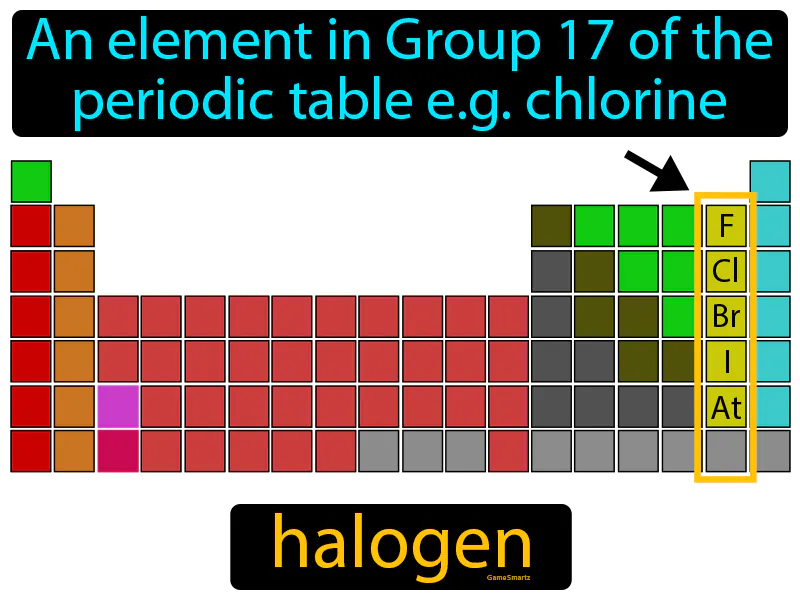
Halogen Meaning

Example:
Imagine you're at a party, trying to find the perfect spot to join a conversation without feeling left out. Halogens, like chlorine, are a bit like that—they're always looking to join in and form bonds with other elements to achieve stability. Just as you search for the right conversation to fit into, halogens eagerly seek out electrons from other atoms to complete their outer shell and reach a stable configuration. This desire to connect and complete makes halogens highly reactive, much like your determination to find your place in the social circle.
Practice Version

Halogen: An element in Group 17 of the periodic table, like chlorine. Halogen. Halogens are reactive nonmetals that can form salts when combined with metals.