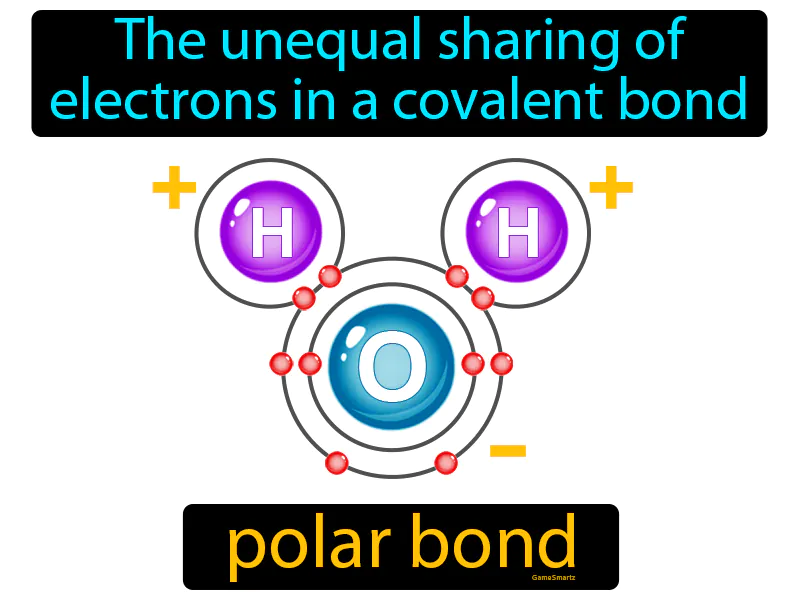
Polar Bond Meaning

Example:
Imagine you're sharing a blanket with someone on a cold night, but your partner keeps hogging most of it. This uneven distribution of the blanket is similar to the unequal sharing of electrons in a polar covalent bond. Just like one person ends up with more of the blanket, in a polar bond, one atom attracts the shared electrons more strongly, creating a partial negative charge, while the other atom becomes partially positive, much like the chilly person left with less blanket.
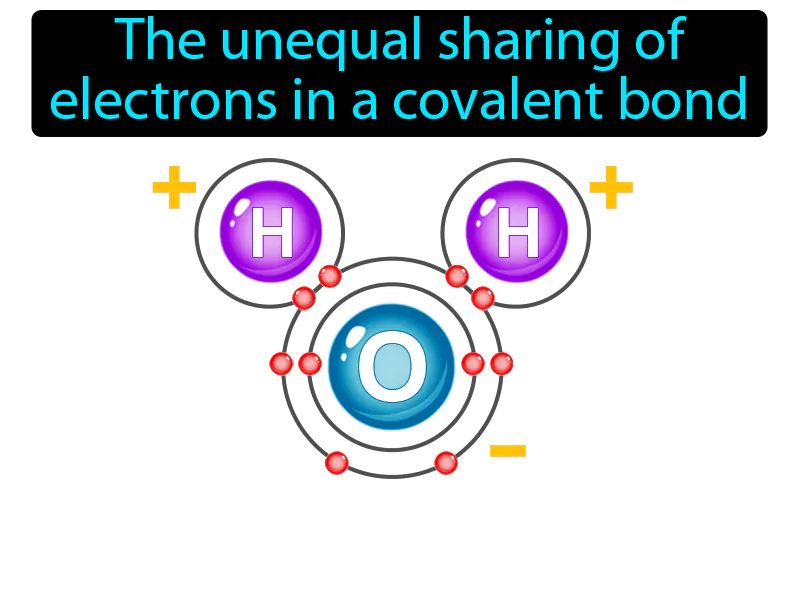
Practice Version
Polar Bond: The unequal sharing of electrons in a covalent bond. Polar bond. A polar bond is a type of covalent bond where electrons are shared unequally, causing one end to be slightly positive and the other slightly negative.