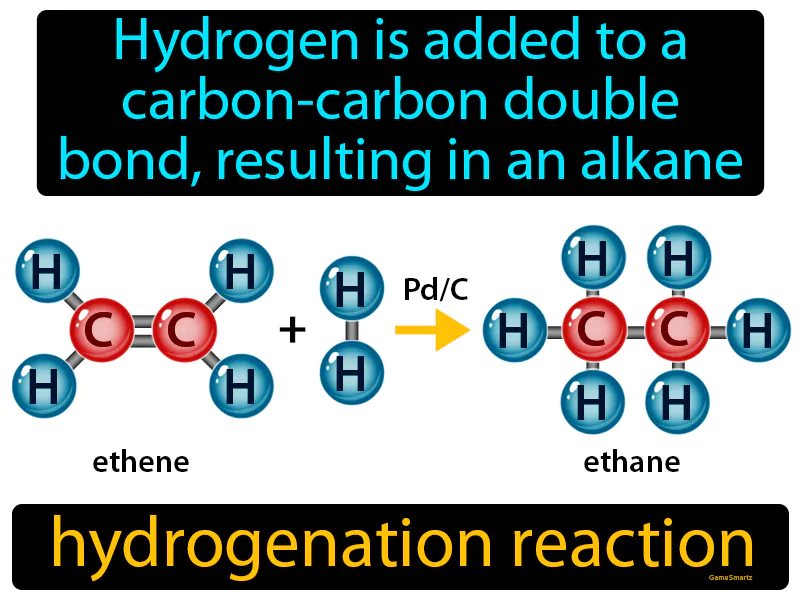
Hydrogenation Reaction
This knowledge is used in careers such as:
Teacher (Teaches Students) ~$55,000 - $95,000 - Number of jobs: 4,700,000 (324,500 Openings Per Year) +2% Growth
Electrical Engineer (Designs Electrical Systems) ~$65,000 - $115,000 - Number of jobs: 295,000 (17,500 Openings Per Year) +7% Growth
Mechanical Engineer (Designs Machines) ~$65,000 - $115,000 - Number of jobs: 290,000 (18,100 Openings Per Year) +9% Growth
Explore These Careers →
Hydrogenation Reaction:
Imagine you're trying to assemble a jigsaw puzzle, but there's a gap right in the middle that makes the picture incomplete. Adding hydrogen to a carbon-carbon double bond in a hydrogenation reaction is like finding the missing puzzle piece that perfectly fits the gap, turning your incomplete picture into a whole, cohesive image. Just as the puzzle piece fills in and completes the picture, hydrogen atoms attach to the double bond, converting it into a single bond and transforming the molecule from an unsaturated alkene to a saturated alkane, completing its structure and stabilizing it.
Practice Version
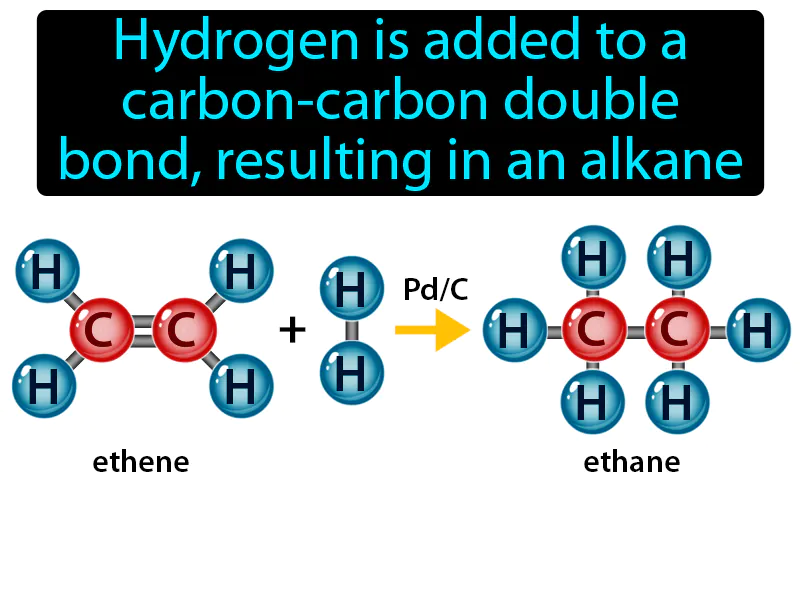
Hydrogenation Reaction: Hydrogen is added to a carbon-carbon double bond, resulting in an alkane. Hydrogenation reaction. A hydrogenation reaction is when hydrogen is added to molecules, often turning unsaturated compounds like oils into saturated ones, like fats.