Aufbau Principle Meaning
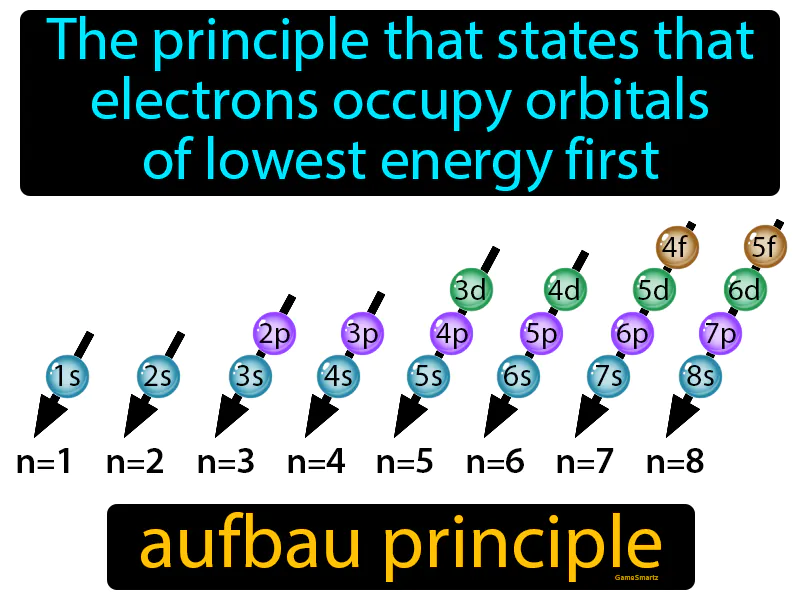
Aufbau Principle:
Imagine you’re at a crowded parking lot, trying to find a spot closest to the entrance. Just like how you'd park in the nearest available spot to minimize the walk, electrons fill the lowest energy orbitals first to minimize energy. The connection here is that both you and the electrons are trying to achieve the most efficient state—whether it’s minimizing your walking distance or the energy of an atom—by occupying the most accessible and advantageous positions available.
Practice Version
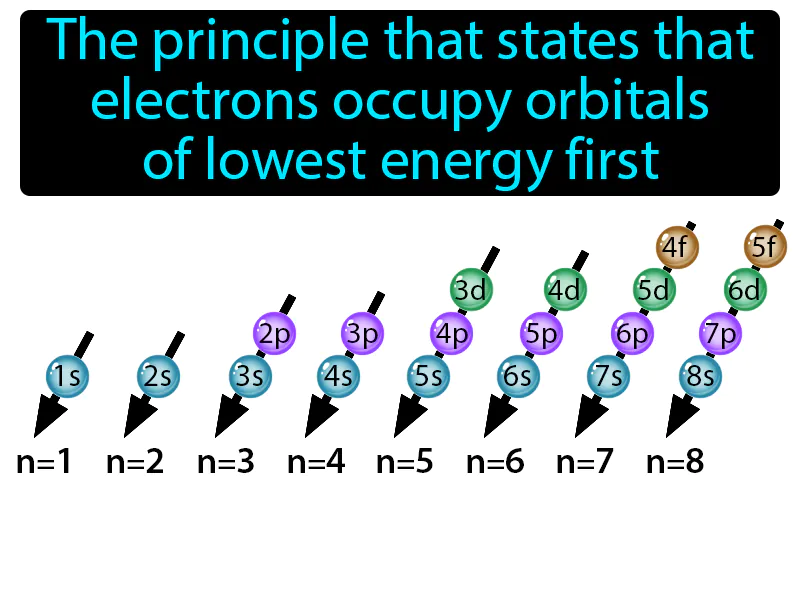
Aufbau Principle: The principle that states that electrons occupy orbitals of lowest energy first. Aufbau principle. The aufbau principle explains how electrons fill the lowest energy levels before moving to higher ones in an atom.