Reversible Reaction Meaning
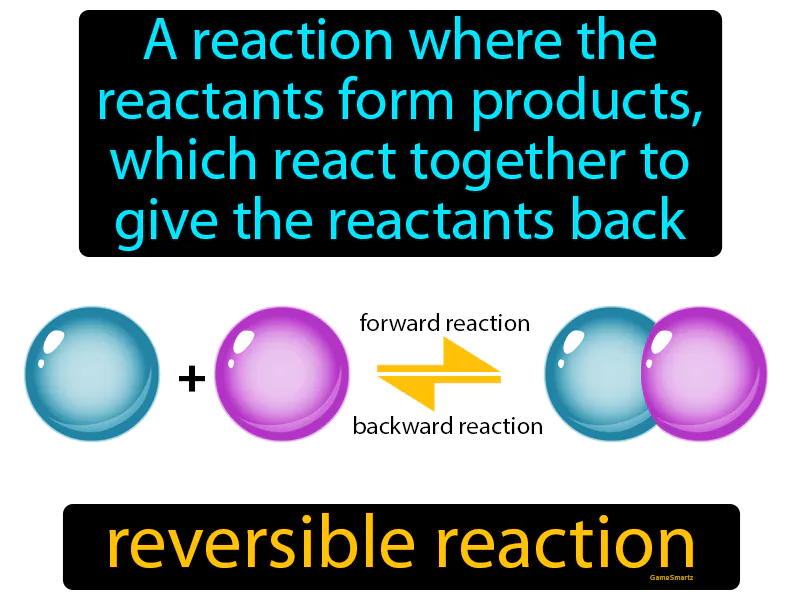
Reversible Reaction:
Imagine trying to decide what to wear in the morning, only to change your mind and switch outfits several times before settling on the first choice. This situation is similar to a reversible reaction, where reactants form products, but under certain conditions, these products can revert to the original reactants. Just like your indecision reflects the back-and-forth nature of reversible reactions, where the "outfits" (products) can go back to being "clothes in the closet" (reactants) until a final balance or equilibrium is reached, your decision-making process mirrors the dynamic balance of chemical reactions.
Practice Version
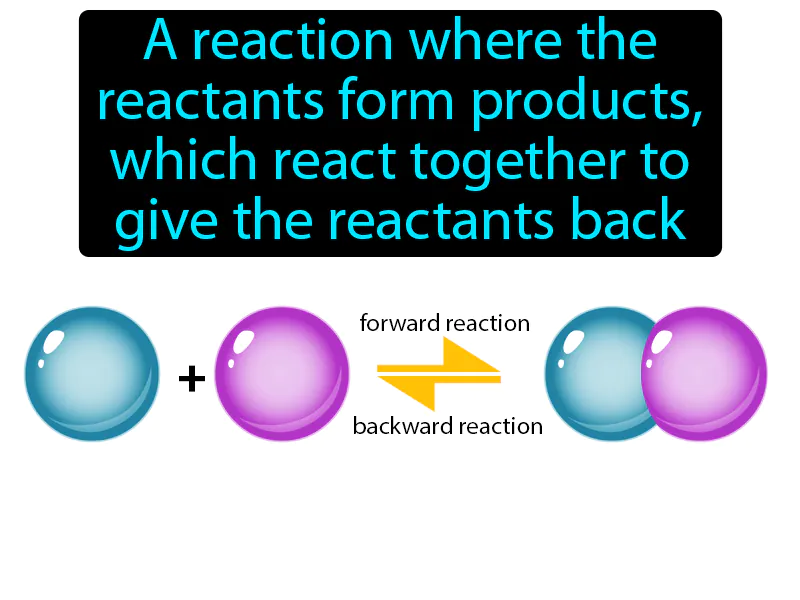
Reversible Reaction: A reaction where the reactants form products, which react together to give the reactants back. Reversible reaction. In Science, a reversible reaction is a chemical process where products can re-form the original reactants.