Atomic Emission Spectrum Meaning
Atomic Emission Spectrum:
Imagine trying to figure out what ingredients are in a mystery soup just by tasting it. Just like how each ingredient in the soup contributes a unique flavor, each element in an atom emits light at specific frequencies, creating a unique pattern. The atomic emission spectrum is like the taste test for light, where scientists "taste" the different frequencies to identify the "ingredients" or elements present, just as you would identify the ingredients in a soup by its flavors.
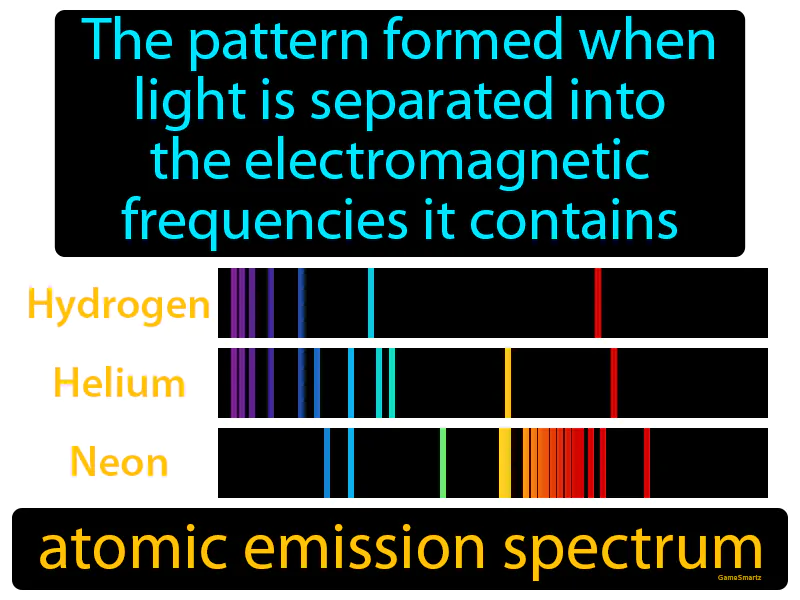
Practice Version
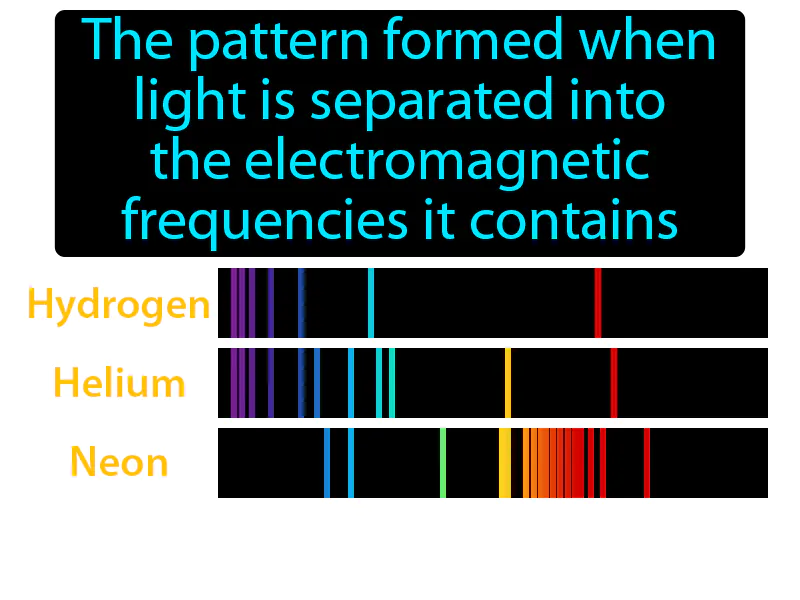
Atomic Emission Spectrum: The pattern formed when light is separated into the electromagnetic frequencies it contains. Atomic emission spectrum. The atomic emission spectrum is a set of bright lines seen when elements emit light at specific wavelengths as electrons jump between energy levels.