Molar Mass Meaning
Molar Mass:
Imagine you're preparing a large batch of cookies and need to know how much flour to use based on the number of cookies you want to make. Just like you need a specific amount of flour to make a set number of cookies, the molar mass tells you how much of a substance in grams is needed to have one mole of its particles. In this analogy, the flour represents the substance, the number of cookies represents the mole, and the recipe's specified amount of flour corresponds to the molar mass, as it ensures you have the right quantity to make the desired number of cookies or moles.

Practice Version
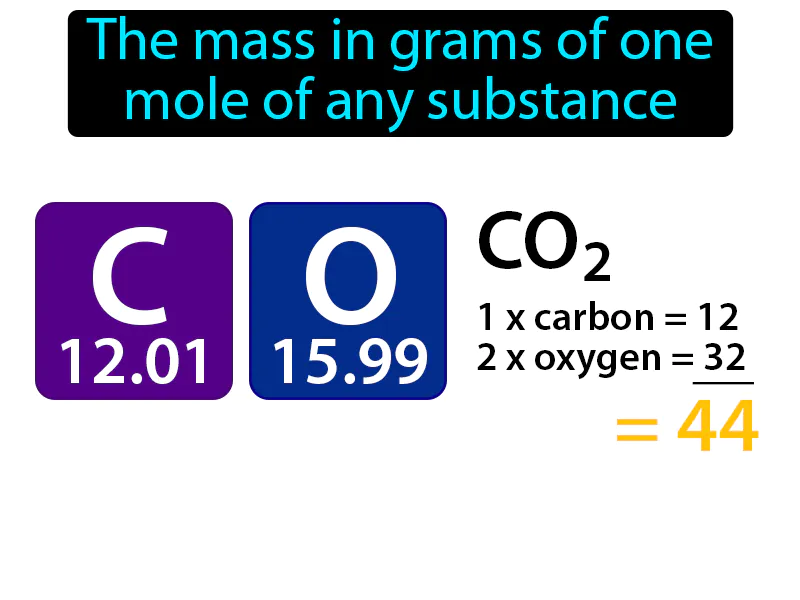
Molar Mass: The mass in grams of one mole of any substance. Molar mass. Molar mass is the weight of one mole of a substance, making it easy to compare how much of one substance is needed relative to another in chemical reactions.