Formula Mass Meaning
Formula Mass:
Imagine you're trying to figure out the cost of making a single sandwich by adding up the prices of each ingredient used. Just like calculating the total price of a sandwich requires summing up the cost of bread, cheese, lettuce, and other ingredients, determining the formula mass of an ionic compound involves adding up the atomic masses of each ion in the compound. In this analogy, each sandwich ingredient represents an ion, and the total sandwich cost represents the formula mass, illustrating how both processes involve compiling individual components to determine a whole.

Practice Version
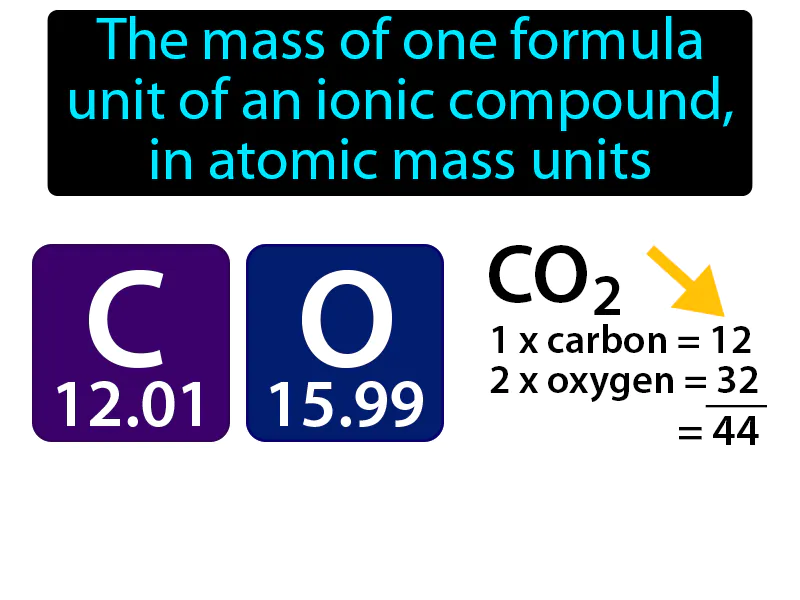
Formula Mass: The mass of one formula unit of an ionic compound, in atomic mass units, is called the formula mass. Formula mass is the total weight of all the atoms in the simplest version of an ionic compound.