Empirical Formula Meaning
Empirical Formula:
Imagine you're trying to split a pizza fairly among several friends, ensuring everyone gets an equal share without any leftovers. Just like dividing the pizza into equal slices, an empirical formula represents the simplest whole-number ratio of atoms, ensuring each element is represented proportionately in the compound. In the same way that you wouldn't cut the pizza into fractions like 1/3 slices for fairness, the empirical formula avoids fractional atoms, presenting the simplest, most balanced representation of the compound's composition.

Practice Version
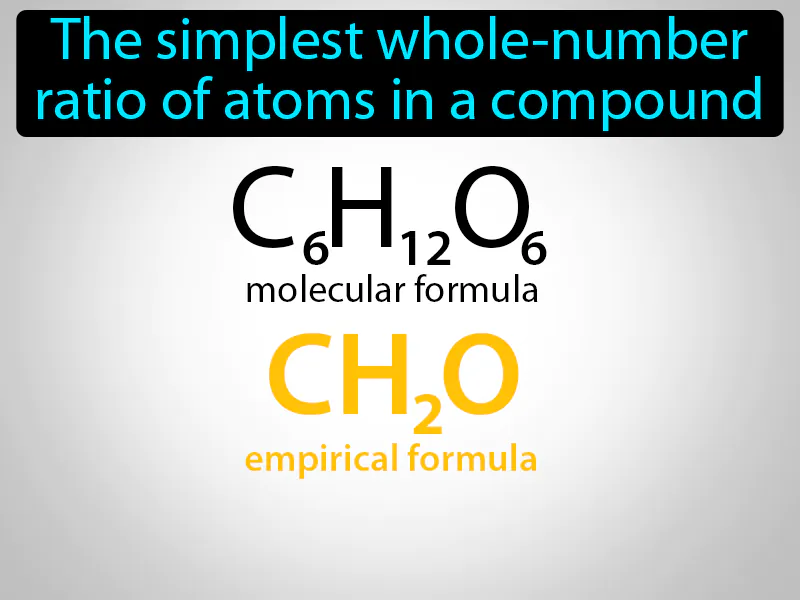
Empirical Formula: The simplest whole-number ratio of atoms in a compound. Empirical formula. An empirical formula shows the smallest ratio of different types of atoms in a compound.