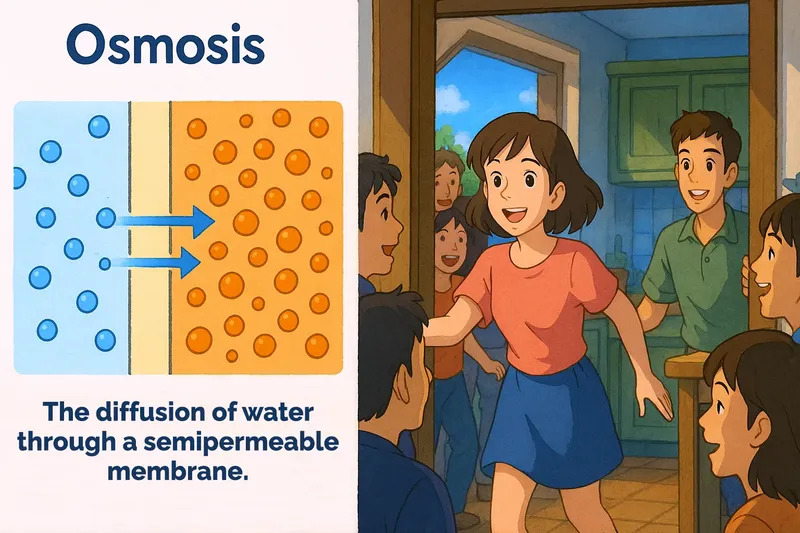
Osmosis Meaning
Example:
Imagine you're at a crowded party and you're trying to move from the packed living room to a less crowded kitchen to get some fresh air. This situation is similar to osmosis, where water moves from an area of high concentration to an area of low concentration through a semipermeable membrane. Just like you moving to a less crowded area for comfort, water molecules move through the membrane to balance concentrations on either side, achieving equilibrium in the process.
Practice Version
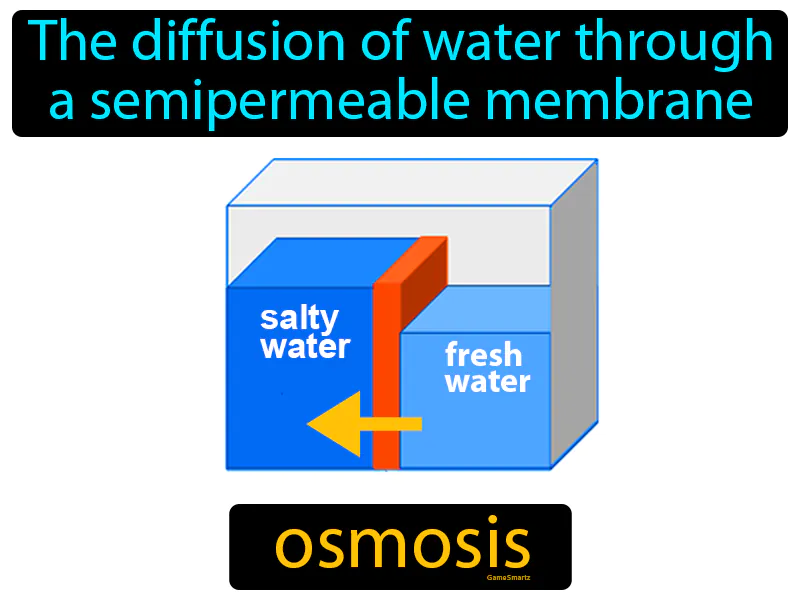
Osmosis: The diffusion of water through a semipermeable membrane. Osmosis. Osmosis is when water moves from a place with more water to a place with less water through a special barrier.