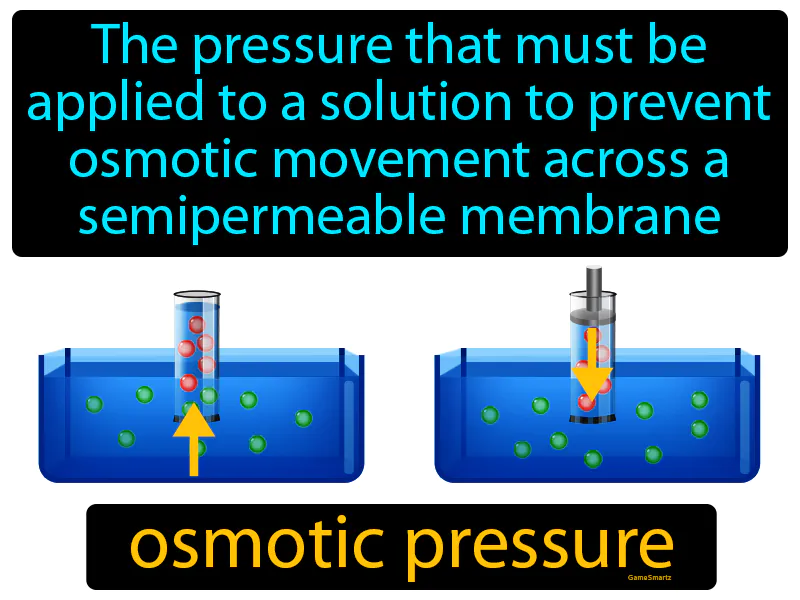
Osmotic Pressure Meaning
Osmotic Pressure:
Imagine trying to stop a crowd of people from pushing through a narrow doorway at a concert. This is similar to osmotic pressure, where you need to apply a force to prevent water molecules from moving through a semipermeable membrane from a less concentrated solution to a more concentrated one. Just as you'd need a security barrier to control the crowd and keep them from moving through the doorway, osmotic pressure acts as the barrier that counteracts the natural tendency of water to flow into the concentrated solution through the membrane.
Practice Version

Osmotic Pressure: The pressure that must be applied to a solution to prevent osmotic movement across a semipermeable membrane. Osmotic pressure. Osmotic pressure is the force needed to stop water from moving through a membrane to balance concentration.