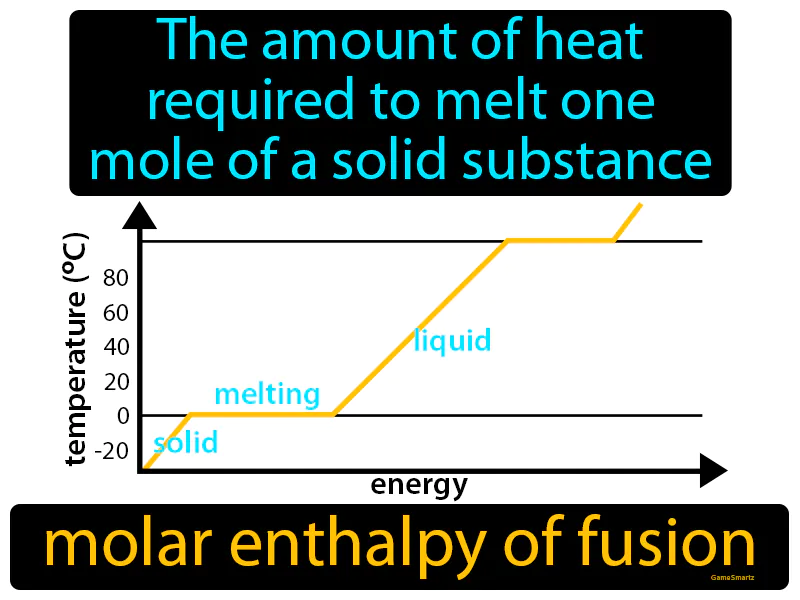
Molar Enthalpy Of Fusion Meaning
Molar Enthalpy Of Fusion:
Imagine trying to get out of bed on a cold winter morning, feeling stuck and frozen under the covers. Just as it takes a certain amount of motivation and energy to break free from the warmth of the blanket and start your day, the molar enthalpy of fusion is the energy needed to transition a mole of a solid substance from its 'stuck' solid state to a more fluid liquid state. In this analogy, the blanket represents the solid structure holding the particles in place, and the motivation or energy you need to get up is akin to the heat required to melt the solid, freeing the particles to move more freely like a liquid.
Practice Version
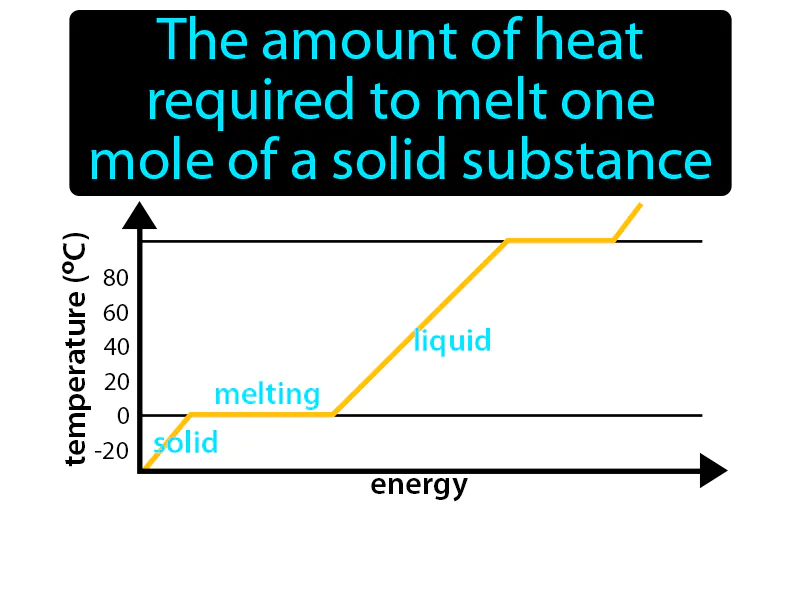
Molar Enthalpy Of Fusion: The amount of heat required to melt one mole of a solid substance. Molar enthalpy of fusion. It is the energy needed to change one mole of a solid into a liquid without changing its temperature.