Hess Law Meaning
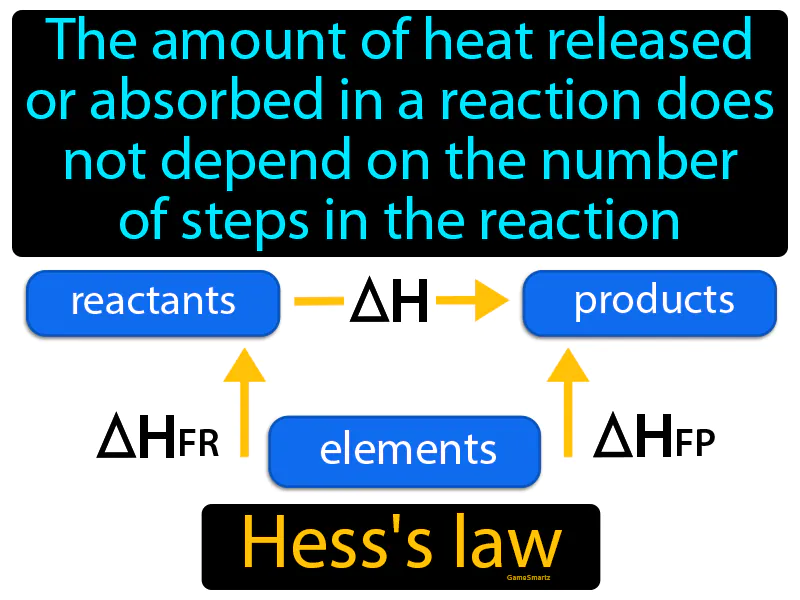
Hess Law:
Imagine you're planning a road trip to a distant city, and you have the option of taking a direct highway or a scenic route with several stops. Regardless of which route you choose, the total distance traveled remains the same. This is similar to Hess's Law, which states that the total heat released or absorbed in a chemical reaction is the same, no matter if the reaction proceeds in one step or multiple steps. Just as the total miles driven are constant irrespective of the path you take, the overall energy change in a chemical reaction is constant, regardless of the reaction pathway.
Practice Version
Hess Law: The amount of heat released or absorbed in a reaction does not depend on the number of steps in the reaction. Hess's law. Hess's law states that the total energy change in a chemical reaction is the same, no matter how many steps the reaction takes.